| |
|
|
Membrane reshaping by Septin filaments
In vivo versus in vitro septin-membrane interaction
Septins form cytoskeletal filaments that assemble at the inner face of the plasma membrane. Septins form rings localized at constriction sites and impact membrane remodeling, Figure 1. Septins are ubiquitous and essential to a variety of cellular processes from cell division, neuron morphogenesis, cell motility, or cellular compartmentalization.
|
| |
|
|
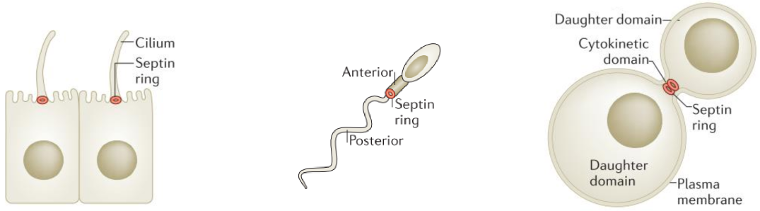
Figure 1.
Septin filament localisations and functions. Septin filaments are localized at constriction sites and impact membrane remodeling (from Caudron et al Dev Cell 2009).
|
| |
|
|
|
|
|
|
|
| |
|
|
At the cleavage furrow, the membrane displays a negative Gaussian
curvature, horse saddle geometry, with both a positive and
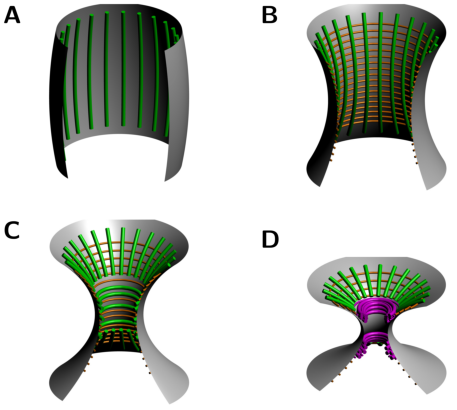
negative curvature. Evidences show that septin filaments globally
reorient by 90° at the onset of cytokinesis in budding yeast, a process known as the Hourglass Model, Figure 2. More specifically, Ong et al (2014) have shown that septin double filaments are
initially arranged radially around the forming bud and progressively align along the mother-bud axis. Then, as the bud neck forms, septin double filaments coexist with septin single filament rings around the bud neck, before reorienting themselve to form two distinct rings aligned circumferentially
on both sides of the bud neck, Figure 3.
|
| |
|
|
Figure 2.
The Hourglass model. (from Ong et al. Nat Commun 2014).
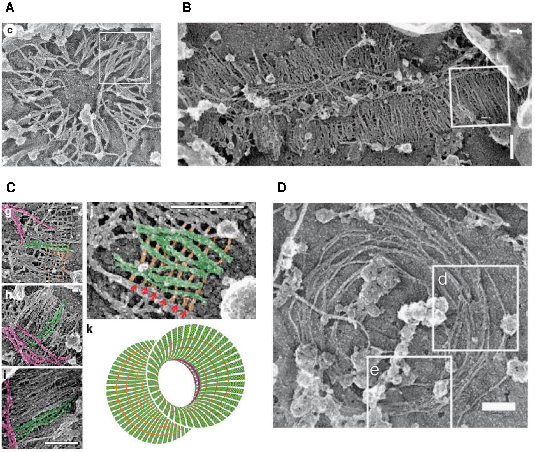
Figure 3.
Experimental evidence. Experimental evidence of the Hourglass to the double ring transition and asynchronous rearrangements of single and double septin filaments at the bud neck. Scale bars 200nm (from Ong et al. Nat Commun 2014).
|
| |
|
|
|
|
|
|
|
| |
|
|
In the experimental part of the project, Aurelie Bertin and Stephanie Mangenot (UMR168) examined, using in vitro tools, how yeast septins behave on curved and deformable membranes. They found that septins reshape the membranes of Giant Unilamellar Vesicles (GUV) with the formation of periodic spikes, Figure 4.
|
| |
|
|
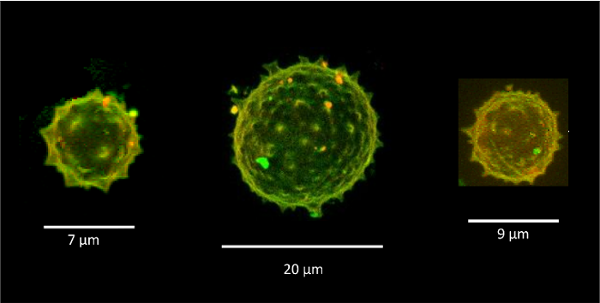
Figure 4.
Septins induce deformations on GUVs.
3D reconstruction of confocal spinning disk images of GUVs in a solution of 600 nM septins (red = lipids, green = septins). Spikes appear on the GUV surface with about 1 μm radius of negative curvature (Beber et al 2019).
|
| |
|
|
|
|
|
|
|
| |
|
|
They also probed how septins bind to bilayers
supported on custom-designed periodic wavy patterns displaying positive and negative micrometric radii of curvatures, Figure 5. Strikingly, septin filaments remain straight and perpendicular to the curvature of the convex parts along the ridges, while bending negatively to follow concave geometries in the valleys, Figure 5.
|
| |
|
|

Figure 5.
Curvature sensitivity of septin filaments.
Septin filaments curve negatively in valleys of custom-made wavy substrates (magenta) while remaining flat and aligned along the ridges (green) (Beber et al 2019).
|
| |
|
|
|
|
|
|
|
| |
|
|
These beautiful images from our experimentalist collaborators caught our curiosity and interest, as no simple interpretation seemed available to explain the stricking sensitivity of septins to negative versus positive micrometric curvatures. Given such micrometric scales, we hypothesized that there might be a generic physical model to account for these observations. Such a model is presented in the next section.
|
| |
|
|
Model of septin-membrane interaction
Curvature sensing is involved in cell and organelle morphology. Four different mechanisms have been proposed to generate and sense membrane curvature. However, in all of these processes, the sensed curvatures are within nanometric range, while septins localize at curvatures of 1 μm-1 as seen around the bud neck during cell division, Figure 3. It is thus crucial to understand how filaments can assemble, grow and organize on micrometric radius of curvatures.
Our main contribution in this project has been to develop a physical model to account for the striking micrometric curvature sensitive adsorption of septin filaments on membranes. The proposed model is intrinsically continuous (filaments are treated as cylinders rather than as a discrete assembly of individual proteins).
Model of septin-membrane interaction on GUV
The first and most direct evidence of septin filament affinity to negative micrometric curvature comes from the observation of GUV deformation, Figure 6, which can be explained with a very simple model,
\(\Delta {g(c)} = \Delta g^{\circ} \Big(1- {1\over 2}\,a\,c\Big) + {1 \over 2} \,kT\, L_{p} {c^2}\)
where \(\Delta g^{\circ}<0\) is the free energy of septin-membrane adsorption, \(a\) the diameter of septin filaments, \(L_p\) their persistence length and \(c\) their local curvature on a membrane, Figure 6. Hence, \(-{1\over 2}\,a\,c\) corresponds to the loss or gain of contact area with the membrane depending on the sign of the curvature \(c\).
This leads to an optimum negative curvature: \(c^\star = 1/R^\star \; =\; \Delta g^{\circ} \,a / 2 kT\, L_{p}\), as shown on GUV, Figure 6.
|
| |
|
|
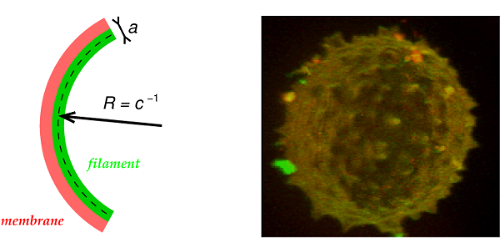
Figure 6.
Model of septin induced deformations on GUVs (Beber et al 2019).
|
| |
|
|
|
|
|
|
|
| |
|
|
This model can be generalized to multiple (\(n\)) septin filaments interacting with a membrane as,
\begin{eqnarray}
\Delta g_n(c) &=& \Delta g^{\circ}_n \Big(1- {1\over 2}\,a_n\,c\Big) + {1 \over 2} \,kT\, L_{p_n} {c^2} \nonumber
\label{1Dequation_multiple_filament}
\end{eqnarray}
with optimum negative curvature of septin multiple (\(n\)) filaments:
\begin{eqnarray}
c_n^\star &=& 1/R_n^\star \; =\; \Delta g^{\circ}_n \,a_n / 2 kT\, L_{p_n} \nonumber
\label{c_nstar}
\end{eqnarray}
where \(a_n\), \(L_{p_n}\), \(\Delta g^{\circ}_n\), \(c_n^\star\) and \(R_n^\star\) are expected to scale as,
\begin{eqnarray}
a_n &\simeq& a_1 \; n^{\delta} \;\;\;\;\;\;\;\;\simeq\;\;\; a_1 \; n^{1/2} \nonumber\\
L_{p_n} &\simeq& L_{p_1} \; n^{4\delta} \;\;\;\;\;\simeq\;\;\; L_{p_1} \; n^{2} \label{persistence_scaling}\nonumber\\
\Delta g^{\circ}_n &\simeq& \Delta g^{\circ}_1 \; n^{1-\delta} \;\simeq\;\;\; \Delta g^{\circ}_1 \; n^{1/2} \nonumber\\
c_n^\star &\simeq& c_1^\star \; n^{1-4\delta} \;\;\;\simeq\;\;\; c_1^\star \; n^{-1} \label{optimumc_scaling}\nonumber\\
R_n^\star &\simeq& R_1^\star \; n^{4\delta-1} \,\,\;\simeq\;\;\; R_1^\star \; n^{1} \label{optimumR_scaling}\nonumber
\end{eqnarray}
assuming a (roughly) circular section of multiple septin filaments (\(\delta=1/2\)).
We then estimated the persistence length(s) of (multiple) septin filaments, using the approach developed for actin filaments in Isambert et al 1995, Figure 7.
|
| |
|
|
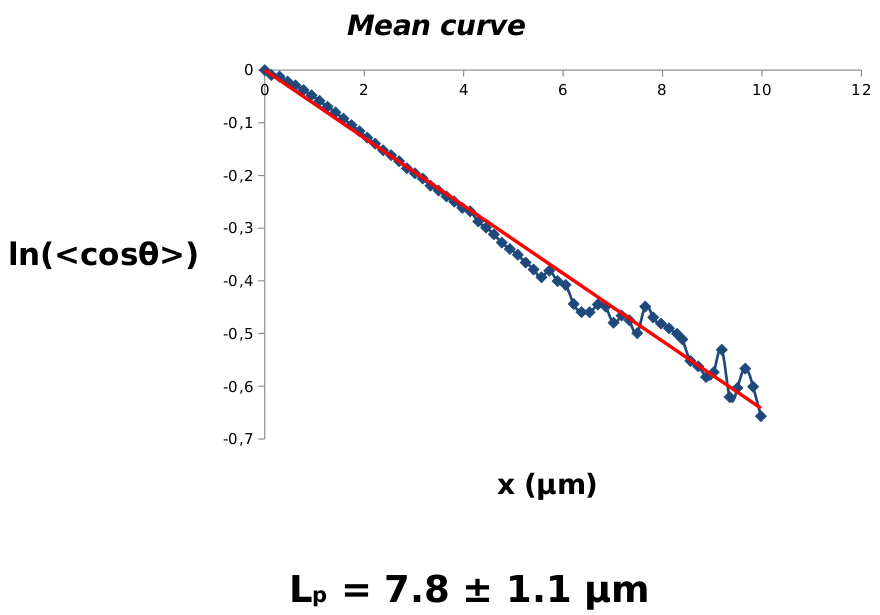
Figure 7.
Persistence length estimate of septin double filaments using the approach developed for actin filaments in Isambert et al 1995.
|
| |
|
|
|
|
|
|
|
| |
|
|
We found: \(L_{p_2}\simeq 8\,\mu\)m and \(R_2^\star \simeq -2.8\,\mu\)m, hence, \(L_{p_1}\simeq 2\,\mu\)m and \(R_1^\star \simeq -1.4\,\mu\)m for optimum radius of negative curvature, in good agreement with observation on GUVs, Figure 4.
It also yields estimates of the minimum radii of curvature for \(n\) filament bundles (corresponding to \(\Delta g_n(c)\simeq 0\)):
\(R^{\rm min}_{n} \simeq \sqrt{kT\, L_{p_n}/ -2\Delta g^{\circ}_n}
\simeq R^{\rm min}_{1} \; n^{3/4}\).
In particular,
\(R^{\rm min}_{1} \simeq 37\) nm, \(R^{\rm min}_{2}\simeq 63\) nm, \(R^{\rm min}_{10}\simeq 208\) nm, \(R^{\rm min}_{30}\simeq 474\) nm and \(R^{\rm min}_{50}\simeq 696\) nm.
|
|
|
|
|
| |
|
|
Flat versus curved filament adsorption models on wavy substrates
The model can be applied to account for the flat versus curved filament adsorption on wavy substrates,
\begin{eqnarray}
\Delta g_n(c)-\Delta g_n(0) &=& -\Delta g^{\circ}_n {1\over 2}\,a_n\,c + {1 \over 2} \,kT\,L_{p_n} {c^2}\nonumber
\label{flat_vs_curved_multiple_filament}
\end{eqnarray}
leading to curved rather than flat adsorptions, \(\Delta g_n(c)-\Delta g_n(0)<0\), for small enough negative curvatures, \(c_n<{\color{red}{c}}<0\), with,
\begin{eqnarray}
c_n &=& 2\,c_n^\star \;=\; \Delta g^{\circ}_n \,a_n / kT\,L_{p_n} \;=\; c_1 \; n^{-1}\nonumber
\label{c_n}
\end{eqnarray}
where \(c_1 = 1/R_1=2\,c_1^\star\) with \(R_1 =R_1^\star/2 \simeq -0.7 \mu\)m and \(c_2 =1/R_2=2\,c_2^\star = c_1/2\) with \(R_2 = 2 R_1 \simeq -1.4 \mu\)m.
Hence, we expect for
\({\color{red}{c}}\lt c_1\lt c_2\lt \cdots\lt c_n\lt 0\) all filaments flat
\(c_1\lt {\color{red}{c}}\lt c_2\lt \cdots\lt c_n\lt 0\) single filaments curved, double filaments flat
\(c_1\lt c_2\lt {\color{red}{c}}\lt \cdots\lt c_n\lt 0\) single and double filaments curved
\(0\lt {\color{red}{c}}\) all filaments flat
This is consistent with the observation of preferential adsorption of septin filaments on wavy substrates, Figure 8.
|
| |
|
|

Figure 8.
Model versus experimental observations.
The model accounts for the observation on wavy substrates, showing that septin filaments curve negatively to maximize their binding energy in valleys (magenta) while they remain flat and align along the ridges (green) to avoid positive curvature with less favorable binding energy (Beber et al 2019).
|
| |
|
|
|
|
|
|
|
| |
|
|
More quantitatively, we expect adsorption of curved single (respectively double) filaments across the groove of undulated membrane surface for \(c_1^*\lt c\lt 0\) (respectively \(c_1^* \lt c_2^* \lt c\lt 0\)), while we expect possible tilted orientations for intermediary negative curvatures \(c_1\lt c\lt c_1^* \lt 0\) (respectively, \(c_2= c_1^* \lt c\lt c_2^* \lt 0\)) in qualitative agreement with experimental observations (see Beber et al 2019).
|
|
|
|
|
| |
|
|
3D Models with axial symmetry
We then considered more biologically relevant 3D models with axial symmetry, Figure 9.
We compared, in particular, radial (\(c_\parallel\)) versus circumferential (\(c_\perp\)) adsorptions of septin filaments with respect to the axis of symmetry.
The basic equations of filament adsorption for this cellular geometry are,
\begin{eqnarray}
\Delta g_n(c_\parallel) &=& \Delta g^{\circ}_n \Big(1- {1\over 2}\,a_n\,c_\parallel\Big) + {1 \over 2} \,kT\, L_{p_n} {c^2_\parallel} \label{2Dequation_multiple_filament_parallel}\nonumber \\
\Delta g_n(c_\perp) &=& \Delta g^{\circ}_n \Big(1- {1\over 2}\,a_n\,c_\perp\,\sin\theta\Big) + {1 \over 2} \,kT\, L_{p_n} {c^2_\perp} \label{2Dequation_multiple_filament_perp}\nonumber
\end{eqnarray}
where \(\theta\) is the angle of the circumferential filament plane with the membrane, Figure 9.
|
| |
|
|
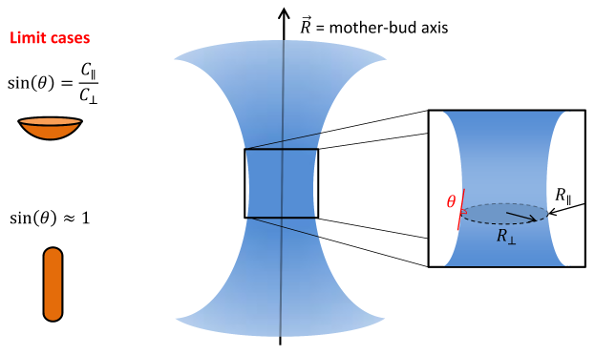
Figure 9.
3D models with axial symmetry (courtesy A. Beber).
|
| |
|
|
|
|
|
|
|
| |
|
|
We considered in more details two limit cases:
1) \(\sin\theta=R_\perp/R_\parallel\) (spherical shape)
2) \(\sin\theta=1\) (oblong shape)
1) Spherical shape
First, for a spherical cell of radius, \(R_\parallel\), we have \(\sin\theta=R_\perp/R_\parallel\) and thus \(c_\perp\,\sin\theta=c_\parallel\) which cancels the difference in area change contributions between radial and circumferential filaments. Hence, we obtain,
\begin{eqnarray}
\Delta g_n(c_\parallel) - \Delta g_n(c_\perp) &=& {1 \over 2} \,kT\, L_{p_n} \Big( {c^2_\parallel} -{c^2_\perp}\Big) \;<\,0 \nonumber
\label{2Dequation_multiple_filament_diff}
\end{eqnarray}
which implies that single or multiple filaments will always adopt a radial orientation, as \(\vert{c_\parallel}\vert < \vert{c_\perp}\vert\), i.e. \(\vert{R_\parallel}\vert > \vert{R_\perp}\vert\), in (locally) spherical geometry, Figure 10, in good agreement with experimental observation (Ong et al 2014).
|
|
|
|
| |
|
|
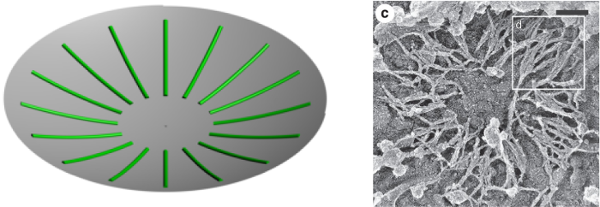
Figure 10.
3D models with axial symmetry.
|
| |
|
|
2) Oblong shape
We then considered a general oblong shape of the (dividing) cell assuming only axial symmetry and \(\theta\simeq \pi/2\) (\(\sin\theta\simeq 1\)) for these oblong shapes,
\begin{eqnarray}
\Delta g_n(c_\parallel) &=& \Delta g^{\circ}_n \Big(1- {1\over 2}\,a_n\,c_\parallel\Big) + {1 \over 2} \,kT\, L_{p_n} {c^2_\parallel} \nonumber\\
\Delta g_n(c_\perp) &\simeq& \Delta g^{\circ}_n \Big(1- {1\over 2}\,a_n\,c_\perp\Big) + {1 \over 2} \,kT\, L_{p_n} {c^2_\perp}\nonumber
\label{2Dequation_multiple_filament_oblong}
\end{eqnarray}
Hence,
\begin{eqnarray}
\Delta g_n(c_\parallel) -\Delta g_n(c_\perp) &\simeq& -{1\over 2}\,\Delta g^{\circ}_n \,a_n \Big(c_\parallel-c_\perp\Big) +{1 \over 2} \,kT\, L_{p_n} \Big( {c^2_\parallel} -{c^2_\perp}\Big) \nonumber
\label{parallel_perp_oblong}
\end{eqnarray}
leading to a transition of septin filament adsorption from parallel (radial) to perpendicular (circumferential) arrangements when the negative parallel curvature, \(c_\parallel<0\), comes closer to zero (i.e. cylinder shape) and ultimately becomes positive, \(c_\parallel>0\), corresponding to a membrane constriction between the mother and daughter cells.
This transition from parallel (radial) to perpendicular (circumferential) adsorptions of septin filaments corresponds to
(with \(c_\parallel-c_\perp>0\)),
\begin{eqnarray}
0&<&\Big({\color{red}{c_\perp+c_\parallel}}-c_n \Big) \Big(c_\parallel-c_\perp\Big)\nonumber \\
0&<& \Big({\color{red}{{1\over R_\perp}+{1\over R_\parallel}}}- {1\over n R_1}\Big)\Big({1\over R_\parallel}-{1\over R_\perp}\Big)\nonumber
\label{parallel_perp_oblong_cond}
\end{eqnarray}
where \(c_n =\Delta g^{\circ}_n \,a_n / kT\, L_{p_n} = c_1 \; n^{-1}\), with \(c_1 = 1/R_1=\Delta g^{\circ}_1 \,a_1 / kT\, L_{p_1}\) and \(R_1 \simeq -0.7 \mu\)m.
Thus, similarly to septin filaments on wavy substrates, we expect for
\({\color{red}{c_\perp+c_\parallel}}\lt c_1\lt c_2\lt \cdots\lt c_n\lt 0\) all filaments parallel
\(c_1\lt {\color{red}{c_\perp+c_\parallel}}\lt c_2\lt \cdots\lt c_n\lt 0\) only single filaments circumferential
\(c_1\lt c_2\lt \cdots\lt c_n\lt {\color{red}{c_\perp+c_\parallel}}\) all filament bundles circumferential
until the maximum curvature (minimum radius) is reached for \(|R_\perp|\simeq 500-700\) nm, as discussed above.
Hence, interpolating the spherical and oblong limit cases recapitulates the Hour-Glass Model, as depicted in Figure 11, in agreement with the experimental evidence in vivo (Ong et al 2014).
|
| |
|
|
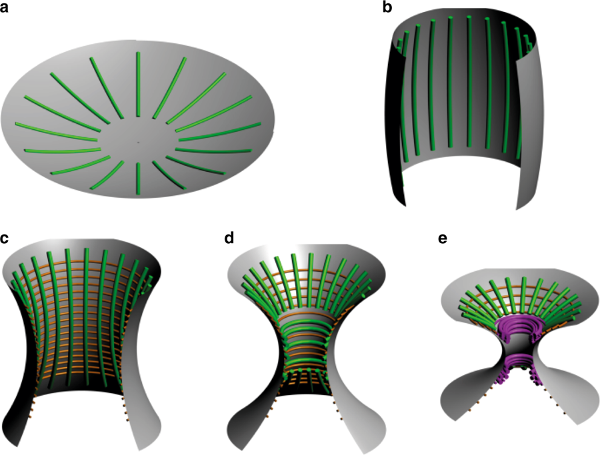
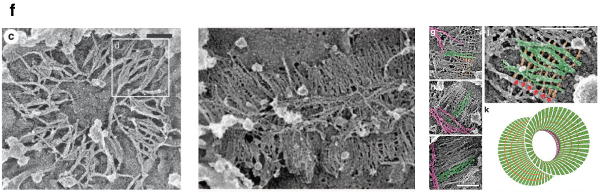
Figure 11.
The Hour-Glass Model. The Hour-Glass Model (Beber et al 2019) and corresponding in vivo experimental evidence (from Ong et al 2014).
|
|
|
|
|
| |
|
|
This minimal theoretical model accounts not only for our
in vitro experimental observations but gives also insights on the
remodeling of the septin filaments in vivo throughout cytokinesis. Indeed
our model explains how the filaments adjust from an
arrangement parallel to the bud axis to a circumferential
arrangement as cytokinesis proceeds. In addition, it suggests that the
bundling of septin filaments might favor their splitting into two
separate ring bundles due to a maximum curvature constraint at the bud neck. Hence, this model suggests that the in situ geometry and
curvature have a role in tuning the organization of septins
in concert with post-translational modifications.
This project has lead to a follow-up paper on the formation of ring-like and tubular structures by septin (Vial et al. 2021).
Finally, this simple generic model may also apply to explain the micrometric curvature affinity of other protein filaments in contact with membranes, such as FtsZ (Osawa et al., EMBO J 2009), MreB (Hussain et al., eLife 2018) and SpoVM (Gill et al., PNAS 2015), or even supra-cellular actin fibers, which organize into thin endodermal versus thick ectodermal perpendicular bundles at organismal level in the hydra (Aufschnaiter et al. Biol Open 2017; Maroudas-Sacks et al. Nat Phys 2021).
|
| |
|
|
|
| |
Related Publications
|
|
Beber A, Taveneau C, Nania M, Tsai F-C, Di Cicco A, Bassereau P, Levy D, Cabral JT, Isambert H, Mangenot S, Bertin A: Membrane reshaping by micrometric curvature sensitive septin filaments.
Nat Commun 10: 420 (2019).
Pubmed
| DOI
| pdf
| supp
Vial A, Taveneau C, Costa L, Chauvin B, Nasrallah H, Godefroy C, Dosset P, Isambert H, Ngo KX, Mangenot S, Levy D, Bertin A, Milhiet PE: Correlative AFM and fluorescence imaging demonstrate nanoscale membrane remodeling and ring-like and tubular structure formation by septins.
Nanoscale 13, 12484-12493 (2021).
Pubmed
| DOI
| pdf
| supp
Isambert H, Venier P, Maggs AC, Fattoum A, Kassab R, Pantaloni D, Carlier MF:
Flexibility of actin filaments derived from thermal fluctuations.
J Biol Chem, 270(19):11437-44 (1995).
Pubmed
| ASBMB
| pdf
|
|














